Valeda Light Therapy
What is Valeda?
Valeda is an innovative treatment that uses precise wavelengths of red and infrared light to address the cellular processes involved in dry macular degeneration (AMD). Through photobiomodulation (PBM), Valeda stimulates mitochondrial activity, supports healthy cell metabolism, and helps reduce inflammation and cell death. This advanced therapy works to slow the progression of dry AMD, improve vision, and even decrease drusen—the small yellow deposits that build up in the retina.
Benefits of Valeda
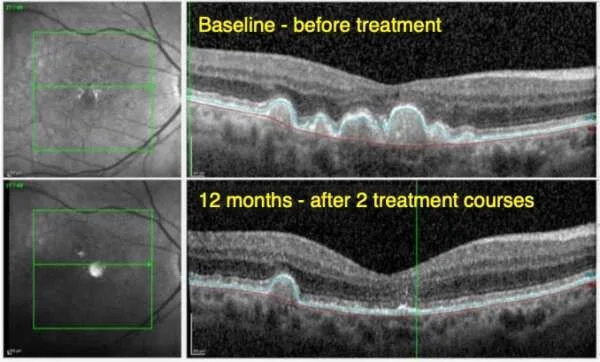
Valeda has delivered remarkable results in clinical trials. In the LIGHTSITE III trial, patients who underwent photobiomodulation (PBM) therapy experienced:
Improved visual acuity, with an average gain of more than one line on the eye chart after one and two years of treatment.
Reduced drusen volume, decreasing the macular deposits associated with vision loss.
Slowed progression of geographic atrophy, with significantly fewer cases of this condition in treated patients compared to those receiving a sham treatment.
These clinical findings are an exciting step forward, offering patients with dry macular degeneration (AMD) a proven option to maintain vision and slow disease progression.
Treatment Details
Valeda uses a cutting-edge light delivery system to emit red and infrared light. The treatment is non-invasive, safe, and performed through multiple sessions over a few weeks.
Treatment Schedule: Most patients complete a series of 9 treatment sessions over 3-5 weeks. After the initial series, follow-up treatments are typically scheduled every 4 months.
No Pain: Valeda is well-tolerated, with most patients reporting no pain or discomfort during the procedure.
FDA Approval: Valeda is the first FDA-approved device for dry macular degeneration (AMD) and the first treatment specifically for intermediate dry AMD. It represents a groundbreaking advancement for patients with this condition.
Is Valeda Right for you?
Valeda is an excellent option for patients with dry macular degeneration (AMD) who have drusen or non-central geographic atrophy. However, this treatment is not recommended for individuals with wet AMD or for those who have advanced vision loss caused by central macular atrophy.
Please contact us today for more information or to schedule a consultation.
Valeda Light Therapy FAQs
-
Valeda is indicated for use in the treatment of dry age-related macular degeneration (AMD) patients with vision impairment. Those individuals receiving treatment should have a doctor diagnosis of Dry AMD. The stage of Dry AMD may impact the benefits of the treatment.
Your doctor will discuss your diagnosis and treatment plan with you.
-
As a precaution, patients have not been tested and should not be treated with Valeda if they have any known reactions to light exposure, or if they have a history of light-activated central nervous system disorders (e.g., epilepsy, migraine). In addition, patients should not receive treatment within 30 days of using any photosensitizing medications (e.g., topicals, injectables) before consulting with their physician.
-
If you wear glasses or contact lenses, you will be asked to remove them prior to receiving treatment. Your eyes will not be dilated for the Valeda treatment. You will be seated comfortably at the Valeda Light Delivery System. The person administering the treatment will guide you through each step of the treatment which takes less than 5 minutes. Treatment for both eyes will take less than 10 minutes.
Clinical trials have shown benefits in visual function following treatment 3 times a week for 3-4 weeks. This is the recommended treatment protocol for patients with Dry AMD. In a recent clinical study, patients received no more than 3 treatments a week and no more than one treatment in a 24 hour period. If you miss a treatment, it is recommended to make up the session within the 3-4 week treatment series period.
-
Treatment with Valeda improves benefits in overall visual function including improvement in vision when scored on the eyechart and improved detailed vision. Photographic imaging also shows improvement in scans of the eye when assessed for drusen (protein deposits that are observed in the diseased eye).
-
Photobiomodulation (PBM) is a low level light based therapy that helps cells improve their function. PBM has been used in many different diseases and disorders for decades with a positive safety profile. In clinical trials in dry AMD patients, the use of light therapy has also been shown to be safe. Valeda is designed to be eye safe. There have been no treatment-related side effects noted in previous studies with the Valeda.
-
It is well known that looking at bright light can produce an afterimage. Another name for this is photobleaching, where the cells that “see” that specific color become fatigued. For example, if you look at a red light, those colors may fade after treatment and you will see more of the opposite color (i.e., green). This phenomenon may be seen immediately following the treatment and will recover in a couple of minutes. You will be able to partake in normal activities following the treatment.
-
Patients can receive the Valeda treatment if they have cataracts.